New Drug Approvals
New Drug Approvals
| 1 | Marzulene S Combination Granules | Gastritis, Gastric ulcer, duodenal ulcer therapeutic agent (1968:Japan, 1990:China, 2000:India) |
| Marzulene Combination Tablets 1.0ES | (Dosage form addition, 2003:Japan) | |
| Marzulene Combination Tablets 0.5ES | (Dosage form addition, 2008:Japan) | |
| Marzulene Combination Tablets 0.375ES | (Dosage form addition, 2009:Japan) | |
| 2 | AZULOXAⓇ Granules 2.5% | Gastric ulcer therapeutic agent (2001:Japan, 2004:Korea) |
| AZULOXAⓇ Tablets 15mg | (Dosage form addition, 2011:Japan) | |
| 3 | SuglatⓇ Tablets 25mg/50mg (Ipragliflozin (ASP1941) ) |
Diabetes therapeutic agent/Selective SGLT2 Inhibitor (Type 2 Diabetes, 2014:Japan, 2015:Korea, 2019:Russia, Joint development with Astellas Pharma Inc.) (Type 1 Diabetes, 2018:Japan, Joint development with Astellas Pharma Inc.) |
| 4 | SUJANUⓇ Combination Tablets (Ipragliflozin/Sitagliptin (MK-0431J) ) |
Type 2 Diabetes therapeutic agent/Selective DPP-4 inhibitor/Selective SGLT2 Inhibitor (2018:Japan, Joint development with Astellas Pharma Inc and MSD.) |
| 5 | XOSPATAⓇ Tablets 40mg (giliteritinib (ASP2215) ) |
Antineoplastic drug/FLT3 Mutation (2018:Japan/US, 2019:Europe/Canada, 2020:Taiwan/Korea/Brazil/Australia, 2021:China/Singapore, Joint development with Astellas Pharma Inc.) |
R&D Pipeline(2025.4)
| 1 | ASP1570 | ①Development stage:Global/Phase Ⅰ ②Target:Cancer ③Development:Astellas |
| 2 | ASP2215 (giliteritinib) | ①Development stage:Global/Phase Ⅲ ②Target:Acute myeloid leukemia ③Development:Astellas |
| 3 | S-556971(KT6-971) | ①Development stage:Japan/Phase Ⅱ, Europe/Phase I ②Target:Dyslipidemia ③Development:Kotobuki |
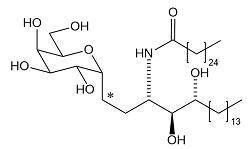
| 4 | KT7-533: α-C-Galactosyl Ceramide
| (Phase I in preparation:Derivation ending) |